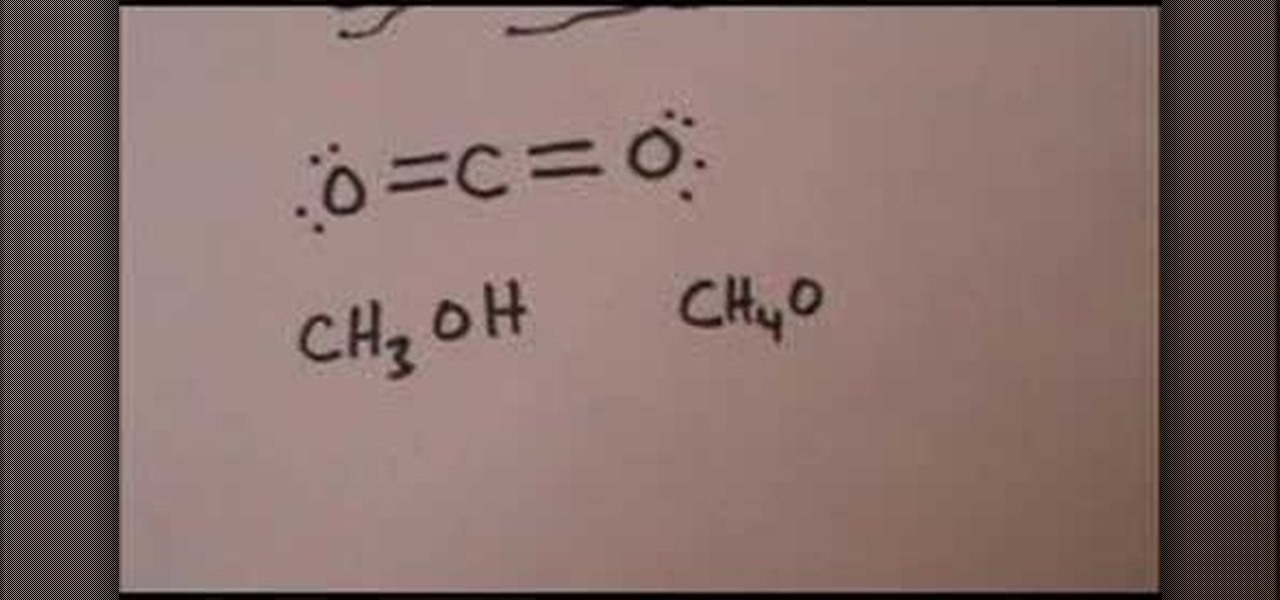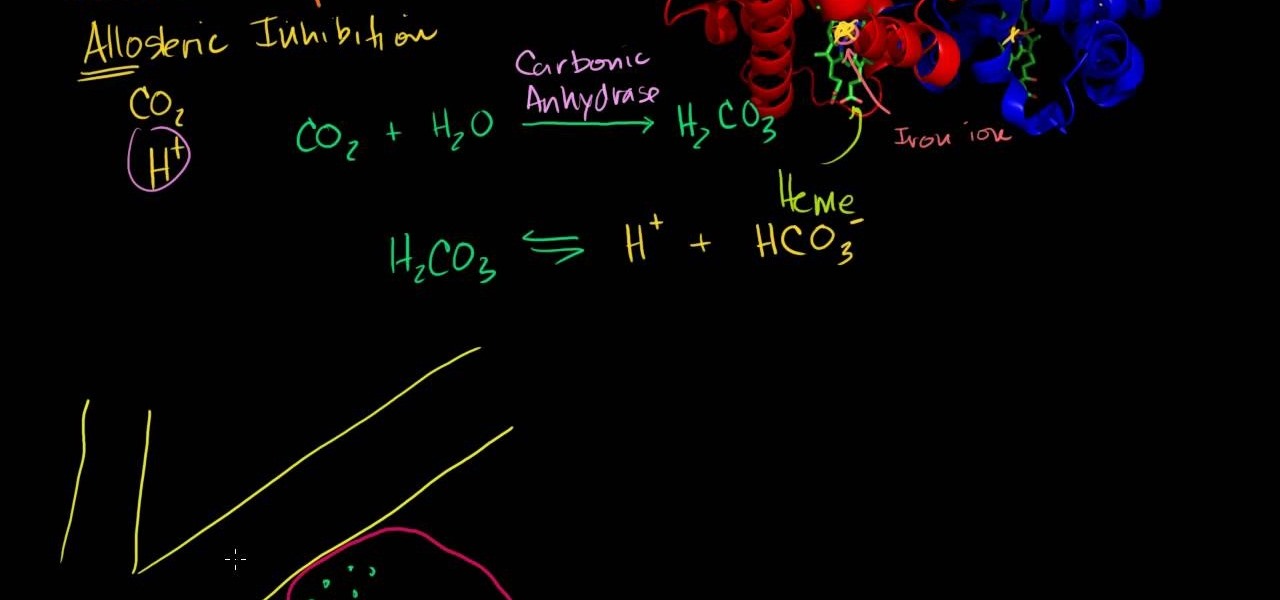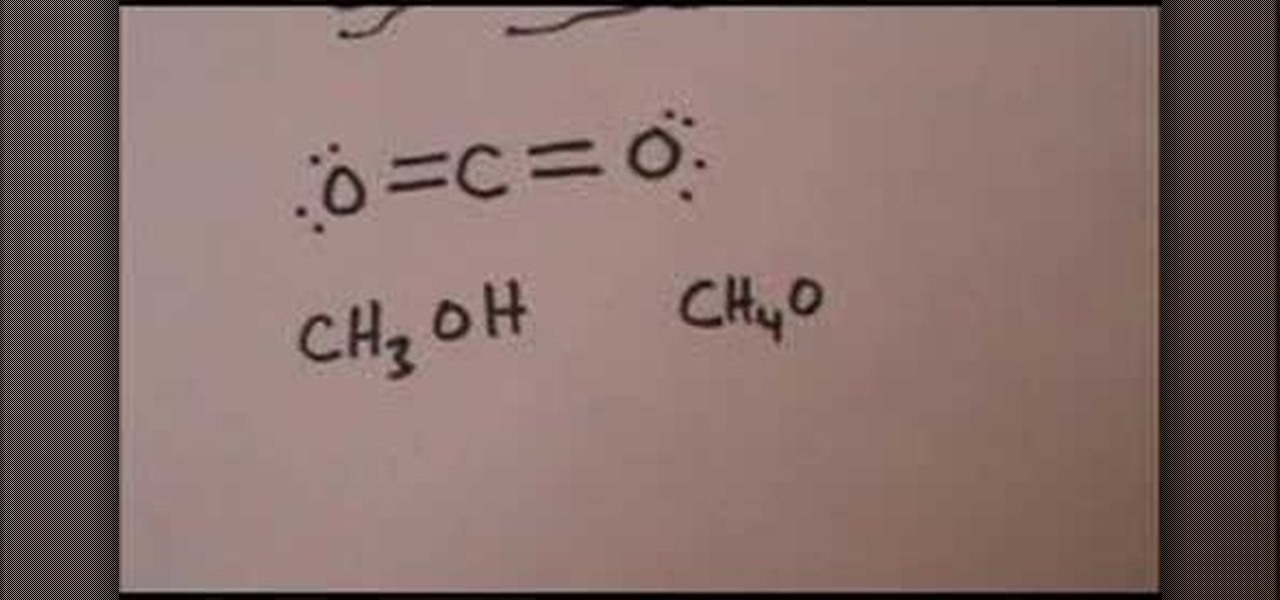
In this Education video tutorial you are going to learn how to draw the Lewis structure for carbon dioxide & methanol. Carbon dioxide is CO2. Oxygen atoms don’t bond together. So, carbon atom has to be in the center. Oxygen has 6 valence electrons and carbon has 4. Hence, carbon wants to form 4 bonds and each oxygen atom wants to form 2 bonds. So, two each electrons of carbon will bond with 2 electrons of each oxygen atom. The Lewis structure for CO2 will be ( …. O=C=O….). The 4 dots within t...

No one ever gets tired of making little explosions by adding vinegar and baking soda. In this video, you'll make a sandwich bomb in a sandwich bag and watch as the contents bubble, brew and explode. Mmmm lunch is ready!

Want to see what happens when baking soda and vinegar become mixed together? Well, this science video tutorial will show you how to do a baking powder and vinegar carbon dioxide experiment.

Mr. G plays with chemicals and fire (what a surprise), for a really cool demonstration. An experiment you'll want to try, and probably can try immediately, as all materials (vinegar and baking soda) are likely in your house already. Exothermic reaction and gas displacement at its best!

In this video tutorial, viewers learn how to use a fire extinguisher. The fire extinguisher contains carbon dioxide. Begin by pulling the pin on the top of the fire extinguisher. The will unlock the lever. Now lift up the nozzle and aim the fire extinguisher at the fire. Then squeeze the lever to release the extinguisher agent. As you are spraying the extinguisher, use a sweeping motion to move it back and forth until the fire is completely out. This video will benefit those viewers who do no...

Watch this science video tutorial from Nurd Rage on how to get zinc, carbon rods and MnO2 from lantern batteries. They show you how to get zinc, carbon electrodes and manganese dioxide from a lantern battery.

In this classic Science Experiment tutorial video, we douse a flame with nothing more than the Carbon Dioxide given off by our homemade Fire Extinguisher.

We all learn a simple description of photosynthesis in grade school but there are a lot more complicated process going on. Plants can utilize carbon at night and this description will explain a very advanced understanding of how CAM works.

C For Chemistry delves into the chemistry of science experiments. This chemist knows what he's talking about. These chemistry experiments are not only fun, but very educational for all of those interested in scientific chemical reactions and properties.

Sure, carbon and oxygen are two of very most fundamental building blocks of all life on Earth — but what have they done for you lately? With this free video guide, you'll learn how to build a safe, simple carbon-dioxide-powered bottle rocket with baking soda and a spent soft plastic waterball.

Various electrochemical reactions requires that anodes do not degrade when used. Carbon is cheap, but degrades easily and platinum is extremely expensive. In a previous video, you learned "How to make cobalt and manganese nitrates", and you saw that titanium could be used as a cathode, but not as an anode due to an effect called passivation.

In this trick, use vinegar and baking soda to isolate clear carbon dioxide gas, which you can then pour onto a candle to magically extinguish it.

Watch this video tutorial to learn how to calculate your carbon footprint. A carbon footprint is an equation that estimates just how much you, personally, are contributing to global warming—and all you have to do is plug some info into an online calculator. Figuring out how to make your footprint smaller, of course, is another matter.

This reaction is between metallic magnesium and carbon dioxide. Magnesium reacts with oxygen in the air to form magnesium oxide, but when the only source of oxygen is from CO2 the reaction becomes much more energetic. The products are white magnesium oxide and black carbon.

In this video, I will show you how to make copper glow red hot with the catalytic oxidation of acetone. For this science experiment, we'll just need some acetone, copper wire, and a flame source to initially heat the copper coil we'll make.

Learn how to proof yeast. Yeast is used in baking as a leavening agent, which is what makes bread and pastries light and airy. Yeast reacts with moisture, sugar, and heat to create tiny bubbles of carbon dioxide which cause the dough to expand and rise. To proof yeast, you'll need yeast, sugar, moisture and warm environment.

This video helps us understand the organic molecules and elimination reaction. Take some sugar in a beaker. Sugar has 12 carbon atoms, 22 hydrogen atoms and 11 atoms of oxygen. The sulphuric acid is poured into the sugar and the color change is observed. The color of the sugar gradually changes into black. The sulphuric acid causes an exothermic reaction which releases a large amount of sulphur dioxide gas. All the water (containing hydrogen and oxygen atoms) is eliminated out of the sugar du...

Sea otters are the largest members of the weasel family. When people started hunting sea otters for their fur, their population fell from roughly 225,000 to about 1,500, until the International Fur Seal Treaty took effect in 1911. Since the international ban on otter hunting, the population has rebounded back to roughly 107,000.

Sick of spending your hard earned money on the bourgeois bubbly water? Why spend extra money on bottled seltzer when you can make it at home? Plus, by using fewer plastic bottles, you can reduce your carbon footprint.

If you have some dry ice, try this cool science experiment out. It's so easy, you have to try it, at least once! You just need a chunk of dry ice and some metal coins, like a half dollar (which is comprised of copper and nickel).

When it comes to global warming, most of us think of carbon dioxide emissions. While carbon dioxide is the most important greenhouse gas, carbon dioxide emissions have stayed constant for the last three years. On the other hand, methane, the second most important gas, has been steadily rising since 2007.

Everyone starts their mornings differently, but one common thread amongst the masses tends to be the consumption of coffee, albeit in many different forms—instant, pre-ground, K-Cups, Nespresso, and so on. And I'm sure that we all have our own preferred methods, as well.

Sulfuric acid is mixed with sugar, which is attacked by the acid. The final products are carbon, water vapor, and sulfur dioxide gas.

A fire snake, also referred to as a black snake or sugar snake, is a classic science experiment you can do right in your own kitchen using a baking soda and sugar mixture and a fuel to ignite the reaction.

What is MnSO4 and MNO2, anyway? They are they molecular formula for Manganese Sulfate and Manganese Dioxide. And you can make one from the other. But how?

Amaze and amuse your friends with this fun magic trick! Using only vinegar and baking soda, which you may remember from some of the bottle-bomb-making videos elsewhere on the site, you can create carbon dioxide which, since it's heavier than air, can be used to invisibly fill a glass and then be poured onto a candle, extinguishing it without looking like it's done anything at all! No word on how long the gas will stay in the glass, so set this one up right before you want to do it and then hi...

Plants all around us capture sunlight every day and convert it to energy, making them a model of solar energy production. And while the energy they make may serve the needs of a plant, the process isn't efficient enough to generate power on a larger scale. So, scientists from the University of California found a way to treat bacteria with chemicals that turned them into photosynthesis machines, capable of generating products we can convert into food, fuels, and plastics.

In this video, we learn how to mow to Make a Dry Ice Crystal Ball! This is a fun activity you can do with your kids and that you will enjoy as well. To start, you will need to take some Dawn dish soap that doesn't have soap and isn't anti-bacterial. After this, put this in water and add rags into it. Swish it around to make the water bubble up. Now, grab a smaller bowl and fill it with water. Then, take another bowl and put dry ice in it using a glove. Next, Now, drag the rag over the top of ...

An in-depth explanation of the structure of the hemoglobin molecule, the process by which it binds with the oxygen in the lungs, how it dumps the same oxygen molecule at the appropriate location in the body and how carbon-dioxide gets attached to the hemoglobin molecule are the topics which the narrator in this video explains with diagrams and a few equations.

This video speaks to everyone who has ever bought anything online, or in fact, anyone who has ever bought anything period. How do you know what you're getting is genuine? Is it a fake product? Is it stolen goods? Is it impure?

Baking powder and baking soda are two staples almost everyone has around that seem to last forever. But a lot of people don't know that they eventually start to lose their potency after enough time on the shelf. If you can't remember when you bought it, it's probably time for a new box.

This actions is a video tutorial in the Education category where you are going to learn how to synthesize copper(II) carbonate & sodium bicarbonate. For this you will need copper sulphate which is available in root kill and sodium bicarb which is baking soda. Take 100g of copper sulphate and dissolve in about 400ml of water. Now take 69.27g of baking soda. Add baking soda very slowly and keep stirring the solution. You got to be very careful as the chemical reaction will produce lot of carbon...

The CVC 3000 is a monitor. It is an active bed bug monitor that use attractants to lure bed bugs to it. It is another tool in bed bug management. It contains a carbon dioxide cylinder/cartridge. The capture slides will be trapped on a tray. The capture slides are one time use capture slides. They have to be replaced each time. The cartridge lasts 10 hours, and it has to be replaced every 10 hours. To use, you will need to place it near the area, where you suspect infestation. Just lie it flat...

In this video Dr. Schultz will show you how to fix stretched or wrinkled earlobes. Here are a few things you can do to remedy stretched or wrinkled earlobes: Laser treatments, especially carbon dioxide treatments, a specified amount of heat is delivered to the skin, and it causes a contraction, which causes a plumping of the earlobe, removing the wrinkles. The results will last for years. With injectable filler treatments like collagen, you will also find a plumping of the skin, but they will...

What's a kegerator, you ask? Why a keg and refrigerator built in one. This short instructional video shows how to find the hot line for the compressor in a home-made keferator. Mount the tap by aligning over the hole and screw it down to the plywood underneath. Carbon dioxide lines run out a hole in the back. Watch this video tutorial and learn how to build a kegerator, keg refrigerator, for your beer out of a Sanyo 4912 fridge.

In this video, I'll be showing you how classic black snakes work and how to make them at home. There are actually two methods covered in the video — one that uses fire and one that does not. So just choose the one that fits best for your situation.

Discover the power of raspberries! Literally. In this NurdRage science video, learn how some crushed raspberries and some titanium dioxide (TiO2) can create a powered dye-sensitized solar cell. Inside a raspberry is something called anthocyanin, which is a flavonoid pigment. Commercially purchased solar cells are way more powerful than this DIY solar cell, but with a little improvement, who knows. Nurd Rage also lets you know how this works.

In order to make manganese dioxide electrodes, you're going to need cobalt nitrate and manganese nitrate to do it. Making cobalt nitrate is fairly easy, but making the manganese nitrate is a little more complicated. But not impossible.

With all due respect to the fine folks at DrinkTanks, it's a little surprising that no one came up with their idea sooner. After all, last I checked, beer was pretty darned popular.

This video shows you how to make bottle of beer appear to instantly freeze. It's hard to believe, but the process is very simple. Take a beer bottle and keep it in refrigerator, do not freeze it. Allow it to cool for some hours. After that take it out and check that it is still in liquid state, then tap it strongly on the bottom side and wait for few minutes. You can see that it has been completely frozen. This is due to the process of carbon dioxide present in the beer bottle.